Phylogenetic branch length distance (BLD): A maximum likelihood (ML) tree is estimated on the MSA of the sequences. The dissimilarity between a pair of sequences is defined as the percentage of non-gapped sites that disagree in the induced pairwise alignment. MSA-based sequence dissimilarity (MSD): All sequences in the data set are aligned into a single MSA. This is the traditional approach for estimating the similarity or dissimilarity between sequences. Pairwise alignment sequence dissimilarity (PSD): For a pair of sequences, an optimal pairwise alignment is computed and the dissimilarity is defined as the percentage of sites that disagree in the pairwise alignment. We will discuss three different dissimilarity metrics for quantifying the evolutionary distance between pairs of sequences: This perspective paper focuses on the problems of using sequence similarity for defining OTUs. found that 18% of the V3 region mapped to two or more rRNA sequences. Even using the hypervariable regions can still lead to ambiguity Huse et al. psychrophilus 11) or the same strain may have multiple copies of the 16S rRNA gene that differ by 5% for some regions (such as Escherichia coli K12 12). For example, two different species may have 99% similar 16S sequences (such as Bacillus globisporus and B. Second, the 97% 16S rRNA sequence similarity threshold used to delineate species is only a rough approximation. 
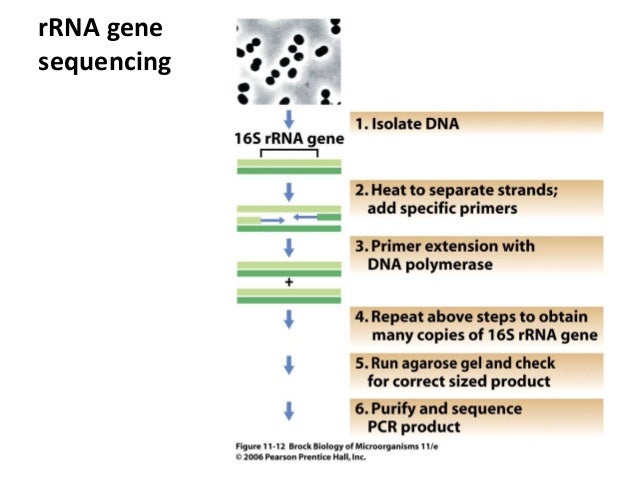
10 These studies suggest that the best practice for computing similarity between sequences is to use evolutionarily corrected distances based upon a MSA however, typical analyses use uncorrected distances based upon pairwise sequence alignments. 9 In addition, the percent similarity is a nonevolutionary-based distance metric it fails to take into account that multiple substitutions can occur at the same site. For example, sequence similarity computed from pairwise alignments underestimates the number of substitutions compared with similarity computed from MSAs. 6– 8 First, the percent sequence similarity can overestimate the evolutionary similarity between pairs of sequences. However, there have been many criticisms with using percent sequence similarity to define OTUs. Thus, clustering allows for rapid analysis of amplicon data sets. Downstream analyses, such as multiple sequence alignment (MSA) or phylogeny estimation, become more tractable when working on the representative sequence set. Typically, a 16S amplicon analysis can have millions of reads, however, this may result in only thousands of OTUs. One of the largest benefits of OTU clustering is computational. Several pipelines have been developed to perform the entire 16S analysis from end to end, including QIIME 4 and MOTHUR. The representative sequence is annotated using a 16S classification method, 2, 3 and all sequences within the OTU inherit that same annotation. 1 From the OTU cluster, a single sequence is selected as a representative sequence. A common similarity threshold used is 97%, which was derived from an empirical study that showed most strains had 97% 16S rRNA sequence similarity. Typically, the similarity between a pair of sequences is computed as the percentage of sites that agree in a pairwise sequence alignment. Sequences are clustered into bins called ‘Operational Taxonomic Units’ (OTUs) based upon similarity. The typical pipeline for 16S amplicon analyses starts with using primers designed to amplify the hypervariable regions of the 16S rRNA gene (typically the V1–V3 region or the V3–V5 region). Although the 16S rRNA gene is highly conserved, there are nine hypervariable regions that can be used to distinguish between different organisms. As the 16S rRNA gene is universally present across bacteria, is highly conserved, and can be easily amplified using universal primers, environmental microbial analyses are often performed using 16S rRNA amplicon sequencing.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
